Endodontic treatment is a predictable procedure with high success rates. Success depends on a number of factors, including appropriate instrumentation, successful irrigation and decontamination of the root-canal space to the apices and in areas such as isthmuses. These steps must be followed by complete obturation of the root canals, and placement of a coronal seal, prior to restorative treatment.
Several irrigants and irrigant delivery systems are available, all of which behave differently and have relative advantages and disadvantages. Common root-canal irrigants include sodium hypochlorite (NaOCl), chlorhexidine gluconate, alcohol, hydrogen peroxide and ethylenediaminetetraacetic acid (EDTA). In selecting an irrigant and technique, consideration must be given to their efficacy and safety.
With the introduction of modern techniques, success rates of up to 98 percent are being achieved.[1] The ultimate goal of endodontic treatment per se is the prevention or treatment of apical periodontitis, such that there is complete healing and an absence of infection,[2] while the overall long-term goal is the placement of a definitive, clinically successful restoration and preservation of the tooth. For these to be achieved, appropriate instrumentation, irrigation, decontamination and root-canal obturation must occur, as well as attainment of a coronal seal.
There is evidence that apical periodontitis is a biofilm-induced disease.[3] A biofilm is an aggregate of microorganisms in which cells adhere to each other and/or to a surface. These adherent cells are frequently embedded within a self-produced matrix of extracellular polymeric substance. The presence of microorganisms embedded in a biofilm and growing in the root-canal system is a key factor for the development of periapical lesions.[4–7] Additionally, the root-canal system has a complex anatomy that consists of arborisations, isthmuses and cul-de-sacs that harbor organic tissue and bacterial contaminants (Figs. 1a,b).[8]
The challenge for successful endodontic treatment has always been the removal of vital and necrotic remnants of pulp tissue, debris generated during instrumentation, the dentin smear layer, microorganisms, and micro-toxins from the root-canal system.[9]
Even with the use of rotary instrumentation, the nickel-titanium instruments currently available only act on the central body of the root canal, resulting in a reliance on irrigation to clean beyond what may be achieved by these instruments.[10] In addition, Enterococcus faecalis and Actinomyces prevention or treatment of apical periodontitis such as Actinomyces israelii — which are both implicated in endodontic infections and in endodontic failure — penetrate deep into dentinal tubules, making their removal through mechanical instrumentation impossible.[11,12] Finally, E. faecalis commonly expresses multidrug resistance,[13–15] complicating treatment.
Therefore, a suitable irrigant and irrigant delivery system are essential for efficient irrigation and the success of endodontic treatment.[16] Root-canal irrigants must not only be effective for dissolution of the organic of the dental pulp, but also effectively eliminate bacterial contamination and remove the smear layer — the organic and inorganic layer that is created on the wall of the root canal during instrumentation. The ability to deliver irrigants to the root-canal terminus in a safe manner without causing harm to the patient is as important as the efficacy of those irrigants.
Over the years, many irrigating agents have been tried in order to achieve tissue dissolution and bacterial decontamination. The desired attributes of a root-canal irrigant include the ability to dissolve necrotic and pulpal tissue, bacterial decontamination and a broad antimicrobial spectrum, the ability to enter deep into the dentinal tubules, biocompatibility and lack of toxicity, the ability to dissolve inorganic material and remove the smear layer, ease of use, and moderate cost.
As mentioned above, root-canal irrigants currently in use include hydrogen peroxide, NaOCl, EDTA, alcohol and chlorhexidine gluconate. Chlorhexidine gluconate offers a wide antimicrobial spectrum, the main bacteria associated with endodontic infections (E. faecalis and A. israelii) are sensitive to it, and it is biocompatible, with no tissue toxicity to the periapical or surrounding tissue.[17] Chlorhexidine gluconate, however, lacks the ability to dissolve necrotic tissue, which limits its usefulness. Hydrogen peroxide as a canal irrigant helps to remove debris by the physical act of irrigation, as well as through effervescing of the solution. However, while an effective anti-bacterial irrigant, hydrogen peroxide does not dissolve necrotic intra-canal tissue and exhibits toxicity to the surrounding tissue.
Cases of tissue damage and facial nerve damage have been reported following use of hydrogen peroxide as a root-canal irrigant.[18] Alcohol-based canal irrigants have antimicrobial activity too, but they do not dissolve necrotic tissue.
embedImagecenter("Imagecenter_1_1324",1324, "large");
The irrigant that satisfies most of the requirements for a root-canal irrigant is NaOCl.[19,20] It has the unique ability to dissolve necrotic tissue and the organic components of the smear layer.[19,21,22] It also kills sessile endodontic pathogens organized in a biofilm.[23,24] There is no other root-canal irrigant that can meet all these requirements, even with the use of methods such as lowering the pH,[25–27] increasing the temperature[28–32] or adding surfactants to increase the wetting efficacy of the irrigant.[33,34] However, although NaOCl appears to be the most desirable single endodontic irrigant, it cannot dissolve inorganic dentine particles and thus cannot prevent the formation of a smear layer during instrumentation.[35]
Calcifications hindering mechanical preparation are frequently encountered in the root-canal system, further complicating treatment. Demineralizing agents such as EDTA have therefore been recommended as adjuvants in root-canal therapy.[20,36] Thus, in contemporary endodontic practice, dual irrigants such as NaOCl with EDTA are often used as initial and final rinses to circumvent the shortcomings of a single irrigant.[37–39] These irrigants must be brought into direct contact with the entire canal-wall surfaces for effective action,[20,37,40] particularly in the apical portions of small root canals.[9]
The combination of NaOCl and EDTA has been used worldwide for antisepsis of root-canal systems. The concentration of NaOCl used for root-canal irrigation ranges from 2.5 to 6 percent, depending on the country and local regulations; it has been shown, however, that tissue hydrolyzation is greater at the higher end of this range, as demonstrated in a study by Hand et al. comparing 2.5 and 5.25 percent NaOCl.
The higher concentration may also favor superior microbial outcomes.41 NaOCl has a broad antimicrobial spectrum,[20] including but not limited to E. faecalis. NaOCl is superior among irrigating agents that dissolve organic matter. EDTA is a chelating agent that aids in smear layer removal and increases dentine permeability,[42,43] which will allow further irrigation with NaOCl to penetrate deep into the dentinal tubules.[44]
General safety precautions
Regardless of which irrigant and irrigation system is employed, and particularly if an irrigant with tissue toxicity is used, there are several general precautions that must be followed. A rubber dam must be used and a good seal obtained to ensure that no irrigant can spill from the pulp chamber into the oral cavity. If deep caries or a fracture is present adjacent to the rubber dam on the tooth being isolated, a temporary sealing material must be used prior to performing the procedure to ensure a good rubber dam seal. It is also important to protect the patient’s eyes with safety glasses and protect clothing from irrigant splatter or spill.
It is very important to note that while NaOCl has unique properties that satisfy most requirements for a root-canal irrigant, it also exhibits tissue toxicity that can result in damage to the adjacent tissue, including nerve damage should NaOCl incidents occur during canal irrigation. Furthermore, Salzgeber reported in the 1970s that apical extrusion of an endodontic irrigant routinely occurred in vivo.[45] This highlights the importance of using devices and techniques that minimize or prevent this. NaOCl incidents are discussed later in this article.
Irrigant delivery systems
Root-canal irrigation systems can be divided into two categories: manual agitation techniques and machine-assisted agitation techniques.[9] Manual irrigation includes positive-pressure irrigation, which is commonly performed with a syringe and a sidevented needle. Machine-assisted irrigation techniques include sonics and ultrasonics, as well as newer systems such as the EndoVac (SybronEndo), which delivers apical negative-pressure irrigation,[46] the plastic rotary F File (Plastic Endo),[47,48] the Vibringe (Vibringe),[49] the Rinsendo (Air Techniques)[9] and the EndoActivator (DENTSPLY Tulsa Dental Specialties).[9]
Two important factors that should be considered during the process of irrigation are whether the irrigation system can deliver the irrigant to the whole extent of the root-canal system, particularly to the apical third, and whether the irrigant is capable of debriding areas that could not be reached with mechanical instrumentation, such as lateral canals and isthmuses. When evaluating irrigation of the apical third, the phenomenon of apical vapor lock should be considered.[50–52]
Apical vapor lock
Because roots are surrounded by the periodontium, and unless the root-canal foramen is open, the root canal behaves like a closed-ended channel. This produces an apical vapor lock that resists displacement during instrumentation and final irrigation, thus preventing the flow of irrigant into the apical region and adequate debridement of the root-canal system.[53,54]
Apical vapor lock also results in gas entrapment at the apical third.[9] During irrigation, NaOCl reacts with organic tissue in the root-canal system, and the resulting hydrolysis liberates abundant quantities of ammonia and carbon dioxide.[55] This gaseous mixture is trapped in the apical region and quickly forms a column of gas into which further fluid penetration is impossible. Extension of instruments into this vapor lock does not reduce or remove the gas bubble,[56] just as it does not enable adequate flow of irrigant.
The phenomenon of apical vapor lock has been confirmed in studies in which roots were embedded in a polyvinylsiloxane impression material to restrict fluid flow through the apical foramen, simulating a closed-ended channel. The result in these studies was incomplete debridement of the apical part of the canal walls with the use of a positive-pressure syringe delivery technique.[57–60]
Micro-CT scanning and histological tests conducted by Tay et al. have also confirmed the presence of apical vapor lock.[60] In fact, studies conducted without ensuring a closed-ended channel cannot be regarded as conclusive on the efficacy of irrigants and the irrigant system.[61–63] The apical vapor lock may also explain why in a number of studies investigators were unable to demonstrate a clean apical third in sealed root canals.[59, 64–66]
In a paper published in 1983 based on research, Chow determined that traditional positive-pressure irrigation had virtually no effect apical to the orifice of the irrigation needle in a closed root-canal system.[67] Fluid exchange and debris displacement were minimal. Equally important to his primary findings, Chow set forth an infallible paradigm for endodontic irrigation: “For the solution to be mechanically effective in removing all the particles, it has to: (a) reach the apex; (b) create a current (force); and (c) carry the particles away.”[67] The apical vapor lock and consideration for the patient’s safety have always prevented the thorough cleaning of the apical 3 mm. It is critically important to determine which irrigation system will effectively irrigate the apical third, as well as isthmuses and lateral canals,[16] and in a safe manner that prevents the extrusion of irrigant.
Manual agitation techniques
By far the most common and conventional set of irrigation techniques, manual irrigation involves dispensing of an irrigant into a canal through needles/cannulae of variable gauges, either passively or with agitation by moving the needle up and down the canal space without binding it on the canal walls. This allows good control of needle depth and the volume of irrigant that is flushed through the canal.[9,63] However, the closer the needle tip is positioned to the apical tissue, the greater the chance of apical extrusion of the irrigant.[67, 68] This must be avoided; were NaOCl to extrude past the apex, a catastrophic accident could occur.[69]
Manual-dynamic irrigation
Manual-dynamic irrigation involves gently moving a well-fitting gutta-percha master cone up and down in short 2- to 3-mm strokes within an instrumented canal, thereby producing a hydrodynamic effect and significant irrigant exchange.70 Recent studies have shown that this irrigation technique is significantly more effective than automated-dynamic irrigation and static irrigation.[9,71,72]
Machine-assisted agitation systems
Sonic irrigation: Sonic activation has been shown to be an effective method for disinfecting root canals, operating at frequencies of 1–6k Hz.[73, 74] There are several sonic irrigation devices on the market. The Vibringe allows delivery and sonic activation of the irrigating solution in one step. It employs a two-piece syringe with a rechargeable battery. The irrigant is sonically activated, as is the needle that attaches to the syringe. The EndoActivator is a more recently introduced sonically driven canal irrigation system.[9,75] It consists of a portable handpiece and three types of disposable polymer tips of different sizes. The EndoActivator has been reported to effectively clean debris from lateral canals, remove the smear layer and dislodge clumps of biofilm within the curved canals of molar teeth.[9]
Ultrasonics: Ultrasonic energy produces higher frequencies than sonic energy but low amplitudes, oscillating at frequencies of 25–30 kHz.[9,76] Two types of ultrasonic irrigation are available. The first type is simultaneous ultrasonic instrumentation and irrigation, and the second type is referred to as passive ultrasonic irrigation operating without simultaneous irrigation (PUI).
The literature indicates that it is more advantageous to apply ultrasonics after completion of canal preparation rather than as an alternative to conventional instrumentation.[9,20,77] PUI irrigation allows energy to be transmitted from an oscillating file or smooth wire to the irrigant in the root canal by means of ultrasonic waves.[9] There is consensus that PUI is more effective than syringe needle irrigation at removing pulpal tissue remnants and dentine debris.[78–80] This may be due to the much higher velocity and volume of irrigant flow that are created in the canal during ultrasonic irrigation.[9,81] PUI has been shown to remove the smear layer; there is a large body of evidence with different concentrations of NaOCl.[9,80–84] In addition, numerous investigations have demonstrated that the use of PUI after hand or rotary instrumentation results in a significant reduction in the number of bacteria,[9,85–87] or achieves significantly better results than syringe needle irrigation.[9,84,88,89]
Studies have demonstrated that effective delivery of irrigants to the apical third can be enhanced by using ultrasonic and sonic devices that demonstrate acoustic micro-streaming and cavitation.[79,81,90,91] Acoustic micro-streaming is defined as the movement of fluids along cell membranes, which occurs as a result of the ultrasound energy creating mechanical pressure changes within the tissue. Cavitation is defined as the formation and collapse of gas and vapor-filled bubbles or cavities in a fluid.
The Apical Vapor Lock theory, proven in vitro by Tay, has been clinically demonstrated[92] to also include the middle third by Vera: “The mixture of gases is originally trapped in the apical third, but then it might grow quickly by the nucleation of the smaller bubbles, forming a gas column that might not only impede penetration of the irrigant into the apical third but also push it coronally after it has been delivered into the canal.” However, more recently Munoz[93] demonstrated that both passive ultrasonic irrigation (PUI) and EndoVac are more effective than the conventional endodontic needle in delivering irrigant to WL of root canals.”
This raises the efficacy question. Two recently published studies examined this issue with both systems by testing their ability to eliminate microorganisms during clinical treatment from infected root canal systems.[94,95] Paiva fund that after a supplementary irrigation procedure using PUI with NaOCl that 23 percent of the samples produced positive cultures. Cohenca’s study examining the clinical efficacy of the EndoVac fund no microbial growth either after post instrumentation irrigation or at the one[92] week obturation appointment.
When questioning these diverse results, one must remember that microbial hydrolysis via NaOCl is an equilibrium reaction. Hand demonstrated that a 50 percent reduction of NaOCl concentration resulted in a 300 percent reduction in dissolution activity.
Accordingly, one must consider both the delivery of the irrigant to full working length, via PUI or apical negative pressure and the total volume of NaOCl exchanged. The volume of an instrumented root canal 19 mm long shaped to a #35 with a 6 percent instrument equals 0.014 cc. Paiva described placement of NaOCl via a NaviTip (Ultradent) at WL — 4 mm during instrumentation and discussed using PUI with #15 K file at WL — 1 mm. Prior to PUI, 2 ml of NaOCl was injected into the canal; however, this could not have filled the apical 4 mm95 due to the apical vapor lock. According to Munoz, the canal was most likely immediately filled with ultrasonically activated NaOCl for one minute,[92] but as just described — only about 0.014 cc would have been effectively available for this exchange and activation. In contrast, the Apical Negative Pressure protocol described by Cohenca et al. approximately 2 ml of NaOCl actively passes through the complete WL for one[92] minute.[96] The difference in volumetric exchange equals 2/0.014 = 14, 200 percent and likely explains the disinfection differential.
The plastic rotary F File
Although sonic or ultrasonic instrumentation is more effective at removing residual canal debris than rotary endodontic files are,[104] and irrigation solutions are often unable to remove this during endodontic treatment, many clinicians still do not incorporate it into their endodontic instrument armamentarium.
The common reasons given for not using sonic or ultrasonic filing are that it can be time-consuming to set up, an unwillingness to incur the cost of the equipment, and lack of awareness of the benefits of this final instrumentation step in endodontic treatment.
It is for these reasons that an endodontic polymer-based rotary finishing file was developed. This new, single-use, plastic rotary file has a unique file design with a diamond abrasive embedded into a non-toxic polymer. The F File will remove dentinal wall debris and agitate the NaOCl without enlarging the canal further.
Pressure-alternation devices
Rinsendo irrigates the canal by using pressure-suction technology. Its components are a handpiece, a cannula with a 7 mm exit aperture, and a syringe carrying irrigant. The handpiece is powered by a dental air compressor and has an irrigation speed of 6.2 ml per minute. Research has shown that it has promising results in cleaning the root-canal system, but more research is required to provide scientific evidence of its efficacy. Periapical extrusion of irrigant has been reported with this device.[101, 102]
The EndoVac apical negative-pressure system
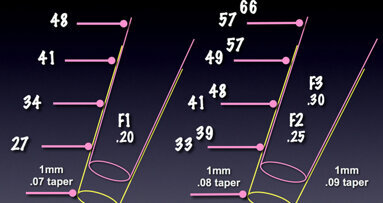
The EndoVac apical negative-pressure irrigation system has three components: the Master Delivery Tip, MacroCannula and MicroCannula. The Master Delivery Tip simultaneously delivers and evacuates the irrigant (Fig. 2). The MacroCannula is used to suction irrigant from the chamber to the coronal and middle segments of the canal. The MacroCannula or MicroCannula is connected via tubing to the high-speed suction of a dental unit. The Master Delivery Tip is connected to a syringe of irrigant and the evacuation hood is connected via tubing to the high-speed suction of a dental unit.56 The plastic MacroCannula has an open end of ISO size 0.55 mm in diameter with a 0.02 taper and is attached to a handpiece for gross, initial flushing of the coronal and mid-length parts of the root canal. The MicroCannula contains 12 microscopic holes and is capable of evacuating debris to full working length.[102]
The ISO size 0.32 mm diameter stainless-steel MicroCannula has four sets of three laser-cut, laterally positioned offset holes adjacent to its closed end, 100 µ in diameter and spaced 100 µ apart. This is attached to a finger piece for irrigation of the apical part of the canal when it is positioned at working length. The MicroCannula can be used in canals that are enlarged with endodontic files to ISO size 35.04 or larger.
During irrigation, the Master Delivery Tip delivers irrigant to the pulp chamber and siphons off the excess irrigant to prevent overflow. Both the MacroCannula and MicroCannula exert negative pressure that pulls fresh irrigant from the chamber, down the canal to the tip of the cannula, into the cannula, and out through the suction hose. Thus, a constant flow of fresh irrigant is delivered by negative pressure to working length. A recent study showed that the volume of irrigant delivered was significantly higher than the volume delivered by conventional syringe needle irrigation within the same period,[46] and resulted in significantly more debris removal at 1 mm from working length than did needle irrigation.
During conventional root-canal irrigation, clinicians must be careful when determining how far an irrigation needle is placed into the canal. Recommendations for avoiding NaOCl incidents include not binding the needle in the canal, not placing the needle close to working length, and using a gentle flow rate when using positive-pressure irrigation.[103] With the EndoVac, in contrast, irrigant is pulled into the canal at working length and removed by negative pressure. Apical negative pressure has been shown to enable irrigants to reach the apical third and help overcome apical vapor lock.[46,104] In addition, with respect to isthmus cleaning, although it is not possible to reach and clean the isthmus area with instruments, it is not impossible to reach and thoroughly clean these areas with NaOCl when the method of irrigation is safe and efficacious. In studies comparing the EndoActivator,[105] passive ultrasonic,[105] the F File,[105] the manual-dynamic Max-i-Probe (DENTSPLY Rinn),105,106 the Pressure Ultrasonic[111] and the EndoVac,[106] only the EndoVac was capable of cleaning 100 percent of the isthmus area.
Apart from being able to avoid air entrapment, the EndoVac system is also advantageous in its ability to deliver irrigants safely to working length without causing their undue extrusion into the periapex,[46,102] thereby avoiding NaOCl incidents. It is important to note that it is possible to create positive pressure in the pulp canal if the Master Delivery Tip is misused, which would create the risk of a NaOCl incident. The manufacturer’s instructions must be followed for correct use of the Master Delivery Tip.
Sodium hypochlorite incidents
Although a devastating endodontic NaOCl incident is rare,[107] the cytotoxic effects of NaOCl on vital tissue are well established.108 The associated sequelae of NaOCl extrusion have been reported to include life-threatening airway obstructions,[109] facial disfigurement requiring multiple corrective surgical procedures,[110] permanent paraesthesia with loss of facial muscle control,[69] and — the least significant consequence — tooth loss.[111]
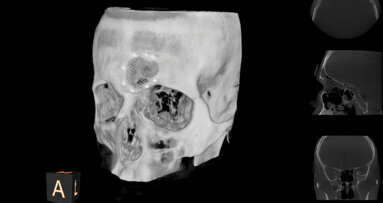
Although the exact etiology of the NaOCl incident is still uncertain, based on the evidence from actual incidents and the location of the associated tissue trauma, it would appear that an intravenous injection may be the cause. The patient shown in Figure 3 demonstrates a widespread area of tissue trauma that is onsistent with the characteristics of NaOCl incident trauma reported by Pashley.[108,112]
This extensive trauma, and particularly involving the pattern of ecchymosis around the eye, could have occurred only if the NaOCl had been introduced intravenously to a vein close to the root apex through which extrusion of the irrigant occurred and the irrigant then found its way into the venous complex. This would require positive pressure apically that exceeded venous pressure (10mg of Hg). In one in-vitro study, which used a positive-pressure needle irrigation technique to mimic clinical conditions and techniques, the apical pressure generated was found to be eight times higher than the normal venous pressure.[113]
This does not imply that NaOCl can or should be excluded as an endodontic irrigant; in fact, its use is critical, as has been discussed in this article. What this does imply is that it must be delivered safely.
Safety first
In order to compare the safety of six current intra-canal irrigation delivery devices, an in-vitro test was conducted using the worst-case scenario of apical extrusion, with neutral atmospheric pressure and an open apex.102 The study concluded that the EndoVac did not extrude irrigant after deep intra-canal delivery and suctioning of the irrigant from the chamber to full working length, whereas other devices did. The EndoActivator extruded only a very small volume of irrigant, the clinical significance of which is not known.
Mitchell and Baumgartner tested irrigant (NaOCl) extrusion from a root canal sealed with a permeable agarose gel.[114] Significantly less extrusion occurred using the EndoVac system compared with positivepressure needle irrigation. A well-controlled study by Gondim et al. found that patients experienced less postoperative pain, measured objectively and subjectively, when apical negative-pressure irrigation was performed (EndoVac) than with apical positive-pressure irrigation.[115]
Efficacy
In vitro and in vivo studies have demonstrated greater removal of debris from the apical walls and a statistically cleaner result using apical negative pressure irrigation in closed root-canal systems with sealed apices. In an in vivo study of 22 teeth by Siu and Baumgartner, less debris remained at 1 mm from working length using apical negative pressure compared with use of traditional needle irrigation, while Shin et al. found in an in vitro study of 69 teeth comparing traditional needle irrigation with apical negative pressure that these methods both resulted in clean root canals, but that apical negative pressure resulted in less debris remaining at 1.5 and 3.5 mm from working length.[46,104,116]
When comparing root-canal debridement using manual dynamic agitation or the EndoVac for final irrigation in a closed system and an open system, it was found that the presence of a sealed apical foramen adversely affected debridement efficacy when manual-dynamic agitation was used, but did not adversely affect results when the EndoVac was used. Apical negative-pressure irrigation is an effective method to overcome the fluid-dynamic challenges inherent in closed root-canal systems.[117]
Microbial control
Hockett et al. tested the ability of apical negative pressure to remove a thick biofilm of E. Faecalis, finding that these specimens rendered negative cultures obtained within 48 hours, while those irrigated using traditional positive-pressure irrigation were positive at 48 hours.[99]
One study found that apical negative-pressure irrigation resulted in similar bacterial reduction to use of apical positive-pressure irrigation and a triple antibiotic in immature teeth.[118] In a study comparing the use of apical positive-pressure irrigation and a triple antibiotic that has been utilized for pulpal regeneration/revascularisation in teeth with incompletely formed apices (Trimix = Cipro, Minocin, Flagyl) versus use of apical negative-pressure irrigation with NaOCl, it was found that the results were statistically equivalent for mineralized tissue formation and the repair process.119 Using apical negative pressure and NaOCl also avoids the risk of drug resistance, tooth discoloration and allergic reactions.[120,121]
Conclusion
Since the dawn of contemporary endodontics, dentists have been syringing NaOCl into the root canal space and then proceeding to place endodontic instruments down the canal in the belief that they were carrying the irrigant to the apical termination.
Biological, scanning electron microscopy, light microscopy and other studies have proven this belief to be in error. NaOCl reacts with organic material in the root canal and quickly forms micro-bubbles at the apical termination that coalesce into a single large apical vapor bubble with subsequent instrumentation. Because the apical vapor lock cannot be displaced via mechanical means, it prevents further NaOCl flow into the apical area.
The safest method yet discovered to provide fresh NaOCl safely to the apical terminus to eliminate the apical vapor lock is to evacuate it via apical negative pressure. This method has also been proven to be safe because it always draws irrigants to the source via suction — down the canal and simultaneously away from the apical tissue in abundant quantities.[122] When the proper irrigating agents are delivered safely to the full extent of the root-canal terminus, thereby removing 100 percent of organic tissue and 100 percent of the microbial contaminants, success in endodontic treatment may be taken to levels never seen before.
Note: This article was published in roots, the international C.E. magazine of endodontics, North America Edition, Vol. 5, Issue 2, 2/2014. This article has been reprinted in part from G. Glassman, Safety and Efficacy Considerations in Endodontic Irrigation (PenWell, January 2011). A complete list of references is available from the publisher.
Over the years, endodontics has diminished itself by enabling the presumption that it is comprised of a narrowly defined service mix; root canal therapy ...
When Dr. Christian Robin and his partners Dr. Benjamin Saleh and Dr. Volong Dao at Saleh Robin & Associates in Ville Mont-Royal, Quebec, were selecting ...
Registration for ROOTS SUMMIT 2024 is open, and the organisers would like to introduce some of the great speakers and their lecture topics for next year’s...
RIDGEFIELD PARK, N.J., USA: DMG has announced that its new generation bulk fill composite and universal bond system has been approved for use in Canada. ...
TORONTO, CANADA: Quantum Dental Technologies (QDT), a medical device company, became the first Canadian business to win the National Instruments (NI) ...
For more than 150 years, Carl Zeiss has developed optical systems that make it possible to see things invisible to the naked eye. For more than 50 years, ...
The concept of “value” has been defined as the worth of a good or service as determined by people’s preferences. And yet with dentists, ...
LONDON, Ontario, Canada: Implants—whether in the jaw or the hip—are a vital part of restoring function and quality of life for millions of patients. ...
The professional, mechanical removal of plaque and biofilm is an integral part of the prophylaxis workflow. The new Proxeo Twist Rotary Polishing System and...
MONTREAL, Quebec, CANADA: At AAE19, the annual session of the American Association of Endodontsts, held April 10 to 13, Sonendo announced it is launching ...
Live webinar
Mon. 8 June 2026
12:00 PM EST (Toronto)
Live webinar
Mon. 8 June 2026
1:00 PM EST (Toronto)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
2:00 PM EST (Toronto)
Live webinar
Wed. 10 June 2026
11:00 AM EST (Toronto)
Live webinar
Wed. 10 June 2026
2:00 PM EST (Toronto)
Nacho Fernández-Baca DDS, MSc
Live webinar
Wed. 10 June 2026
7:00 PM EST (Toronto)
Live webinar
Thu. 11 June 2026
1:00 PM EST (Toronto)



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 International / International
International / International
 Brazil / Brasil
Brazil / Brasil
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East











































To post a reply please login or register